Why Do Nonmetals Have High Ionization Energy
Module Creative Commons License. Poor electrical and thermal conductors.

Do Nonmetals Have High Ionization Energy
The n th ionization energy of an atom is the energy required to detach its n th electron after the first n 1 electrons have already been detached.

. The amount of energy required to add an electron to an atom is called the electron affinity of that atom. Many ionic crystals also have high melting points. Although they are hard they also tend to be.
Common Core Learning Standards. This is due to the very strong attractions between the ionsin ionic compounds the attractions between full charges are much larger than those between the partial charges in polar molecular compounds. This will be looked at in more detail in a later discussion of lattice energies.
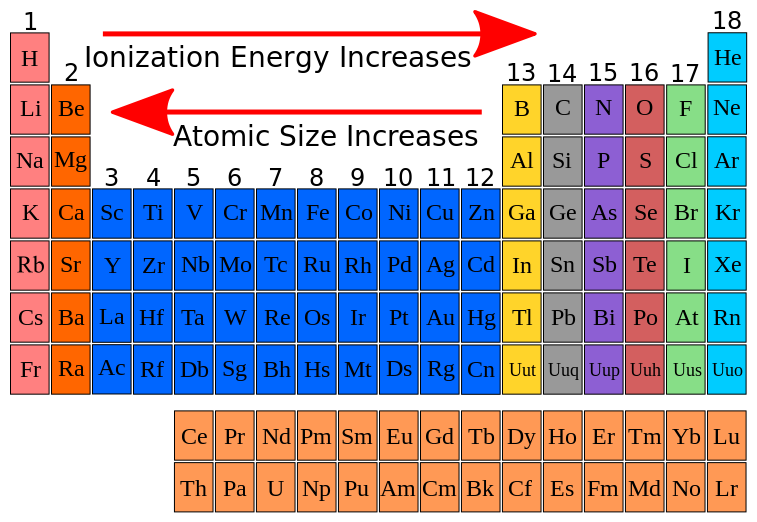
FlexBook Platform FlexBook FlexLet and FlexCard are registered trademarks of CK-12 Foundation. Across a Group on moving top to bottom in a group ionisation energy gradually decreases as atomic radius increases. Common Core Learning Standards CCLS Math.
Little if any metallic luster. In other words electron. SID1 SID2 SID3 SID5 SID6 SID7 SID8 SID9 Resource Type.
The halogens and noble gases are nonmetals although they have their own groups too. The halogens exhibit different physical properties from each other but do share chemical properties. High School Commencement Level Grade 9 Topics.
Represent data with plots on the real number line dot plots histograms and box plots. CCLS State Standard. That is why noble gases also show exceptionally high ionisation energies.
The energy required to detach an electron in its lowest energy state from an atom or molecule of a gas with less net electric charge is called the ionization potential or ionization energy.
Why Is The First Ionization Energy Of A Nonmetal Significantly Higher Than That Of An Alkali Metal Socratic

Explained Why Do Non Metals Have High Ionization Energy

Bell Ringer 1 What Is Ionization Energy Ppt Video Online Download
Comments
Post a Comment